Tobias Krämer
DPhil student
E-mail: tobias.kraemer@chem.ox.ac.uk Dipl.-Chem. University of Hamburg , Germany. Research interests of Tobi:
Publications:Probing the Intrinisic Structure and Dynamics of Aminoborane Coordination at Late Transition Metal Centers: Mono(σ-BH) Binding in [CpRu(PR3)2(H2BNCy2)]+, On the Structural and Electronic Properties of [Zn2(4,4'-bipyridine)(mes)4]n (n = 0-2), a Homologous Series of Bimetallic Complexes bridged by Neutral, Anionic and Dianionic 4,4'-Bipyridine, Exchange coupling through diamagnetic [Fe(CO)4]2- bridging ligands in a xenophilic cluster, Dioxygen Activation by mixed-valent dirhodium complexes of redox non-innocent azoaromatic ligands, N. D. Paul, T. Krämer, J. E. McGrady, S. Goswami Chem. Comm., 2010, 46, 7124. Experimental and Computational Study of the Structural and Electronic Properties of FeII(2,2'-bipyridine)(mes)2 and [FeII(2,2'-bipyridine)(mes)2]-, a Complex containing a 2,2'-Bipyridyl Radical Anion, M. Irwin, R. K. Jenkins, M. S. Denning, T. Krämer, F. Grandjean, G. J. Long, R. Herchel, J. E. McGrady, J. M. Goicoechea Inorg. Chem., 2010, 49, 6160. (H2O)10 and (H2O)12 on a Virtual Metal Surface: The Growth of Ice, H. Henschel, T. Krämer, T. Lankau Langmuir, 2006, 22, 10942. Divergent Synthesis and Biological Evaluation of Carbocyclic α-, iso- and 3'-epi-Nucleosides and their Lipophilic Nucleotide Prodrugs, O. R. Ludek, T. Krämer, J. Balzarini, C. Meier Synthesis, 2006, 8, 1313.
Ferrocenyl-substituted fluorescent anthracenes and anthraquinones, I. R. Butler, A. G. Callabero, G. A. Kelly, J. R. Amey, T. Kraemer, D. A. Thomas, M. E. Light, T. Gelbrich, S. J. Coles Posters and Talks:SWCC 2010: Annual meeting of South West Computational Chemists, 23 September 2010, Bristol, UK. 8th European Conference on Computational Chemistry, 25-28 August 2010, Lund, Sweden. Faraday Discussion 148: Spectroscopy, Theory and Mechanism in Bioinorganic Chemistry, 5-7 July 2010, Nottingham, UK. 42nd IUPAC Congress, 2-7 August 2009, Glasgow, UK. 3rd Scotchem Computational Chemistry Symposium, 13 May 2009, Edinburgh, UK. Current projects:
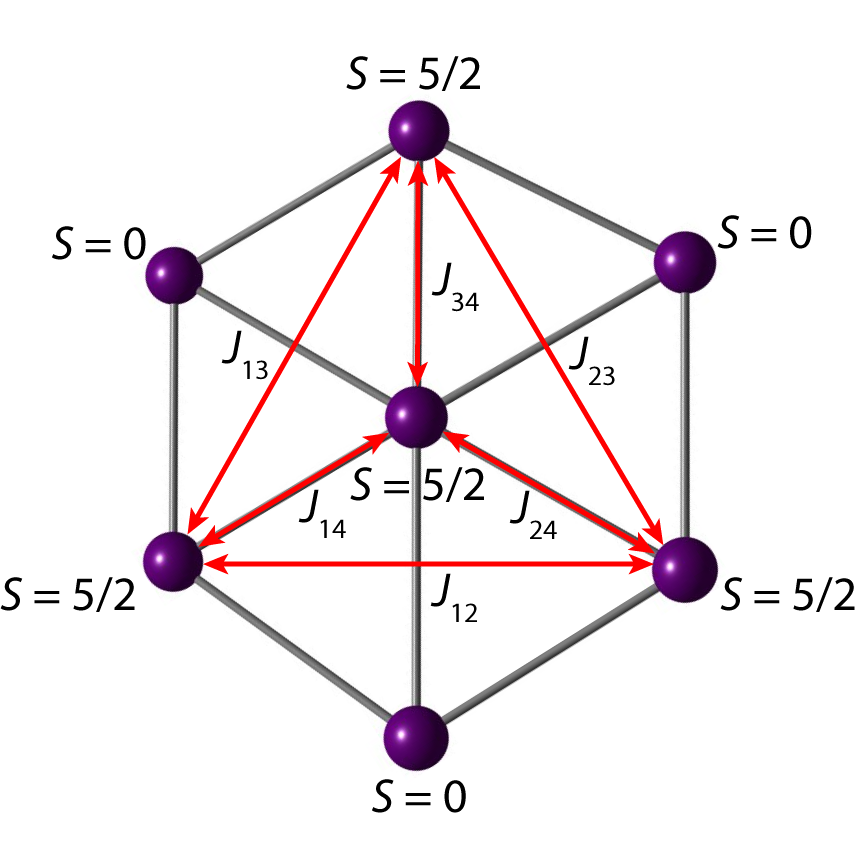
The chemistry of transition metal complexes containing redox-active ligands has experienced a renaissance of research interest, owing to their role as key intermediates in chemical and enzymatic catalysis. In collaboration with Dr. Jose M. Goicoechea (University of Oxford) and Prof. Sreebrata Goswami (Indian Association for the cultivation of Science, Kolkata) we conduct combined experimental and computational studies on transition metal complexes containing non-innocent ligands, such as bipyridine and azoaromatic systems. We use DFT methods to assign the correct electronic ground state and predict spectroscopic and magnetic properties of the investigated complexes. The calculations prove to be a useful tool to support the experimental data from X-ray crystallography, Mössbauer and EPR spectral measurements and help to rationalize the complex magnetic and spectroscopic behaviour of the complexes. Xenophilic clusters comprise metal complex fragments of the two known categories, in which the metal centres are either coordinated by π-acceptor or -donor ligands. We aim to describe the electronic structure and bonding situation within Mn2 and Mn7 clusters, addressing the question of how effective exchange coupling is communicated between the metal centres. The broken symmetry approach is employed to compute coupling parameters Jij from which then magnetic moments are derived by the van Vleck formalism. Different ligands are probed with respect to their ability to communicate superexchange.
Iron containing enzymes play a fundamental role in numerous biological transformations. Among these enzymes, the mono-nuclear non-heme iron oxygenases catalyze a diverse range of oxidative tranformations. The activation of O2 or its derivatives, both of which are used as oxidants, occurs as a crucial step within the catalytic cycle. The involvement of the coordinated ligand is often observed in these oxidation or oxygenation reactions, which include oxygen-atom insertion into aliphatic and aromatic C-H bonds, as well as oxidative dealkylation. An unprecedented case of regiospecific N-oxygenation has been observed in a biomimetic model complex of the non-heme genre. In collaboration with Prof. Christine McKenzie (University of Southern Denmark) we investigate the possible steps involved in this reaction in order to specify the factors that control the unreckoned stereoselectivity, which has been observed experimentally.
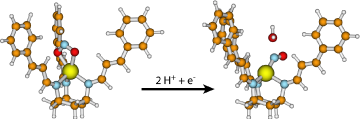
Dissimilatory denitrification represents an integral part of the global nitrogen cycle. Within the denitrification cascade the stepwise reduction of nitrate or nitrite is achieved by prokaryotic organisms to finally produce dinitrogen. Copper Nitrite Reductase (CuNiR) is a key enzyme in the denitrification process, as it catalyses the single-electron reduction of nitrite to nitric oxide, which is the first gaseous product. The catalytic mechanism of CuNiRs remains controversial due to the paradox between observed O-coordination of nitrite and the N-coordination of metal-nitrosyls characterized chemically. The exact function of conserved amino acid residues near the catalytic centre, especially of a hydrophobic isoleucine (Ile257), also remains uncertain. Functional and structural model complexes of the type-2 copper site of CuNiR based on the triaminocyclohexane (tach) ligand system have been designed by our collaborator Prof. Paul Walton (University of York) and his co-workers. The complexes can be derivatised to provide mimics of the immediate and secondary coordination spheres of the active site in CuNiR. Using DFT, we study the details of the electronic structure and mechanistic pathways of nitrite reduction by the above copper(II) complex system.
|